When it comes to software validation ISO 13485:2016 says:

Why you need to validateīoth ISO 13485 and 21 CFR Part 820 are giving a framework for your Quality Management System from a regulatory point of view. It also includes proprietary software, which you create yourself to be more productive. It does not even stop here as you might also use software for human resources, and other parts of the company which by default fall under these requirements. MatrixALM) as well as other software used during development (e.g. MatrixQMS), the documentation of the medical device itself (e.g. You need to validate software used in quality systems, this covers software to manage the QMS itself (e.g. General Principles of Software Validation Final Guidance for Industry and FDA Staff What you need to validate There are more and other methods you can use, e.g.

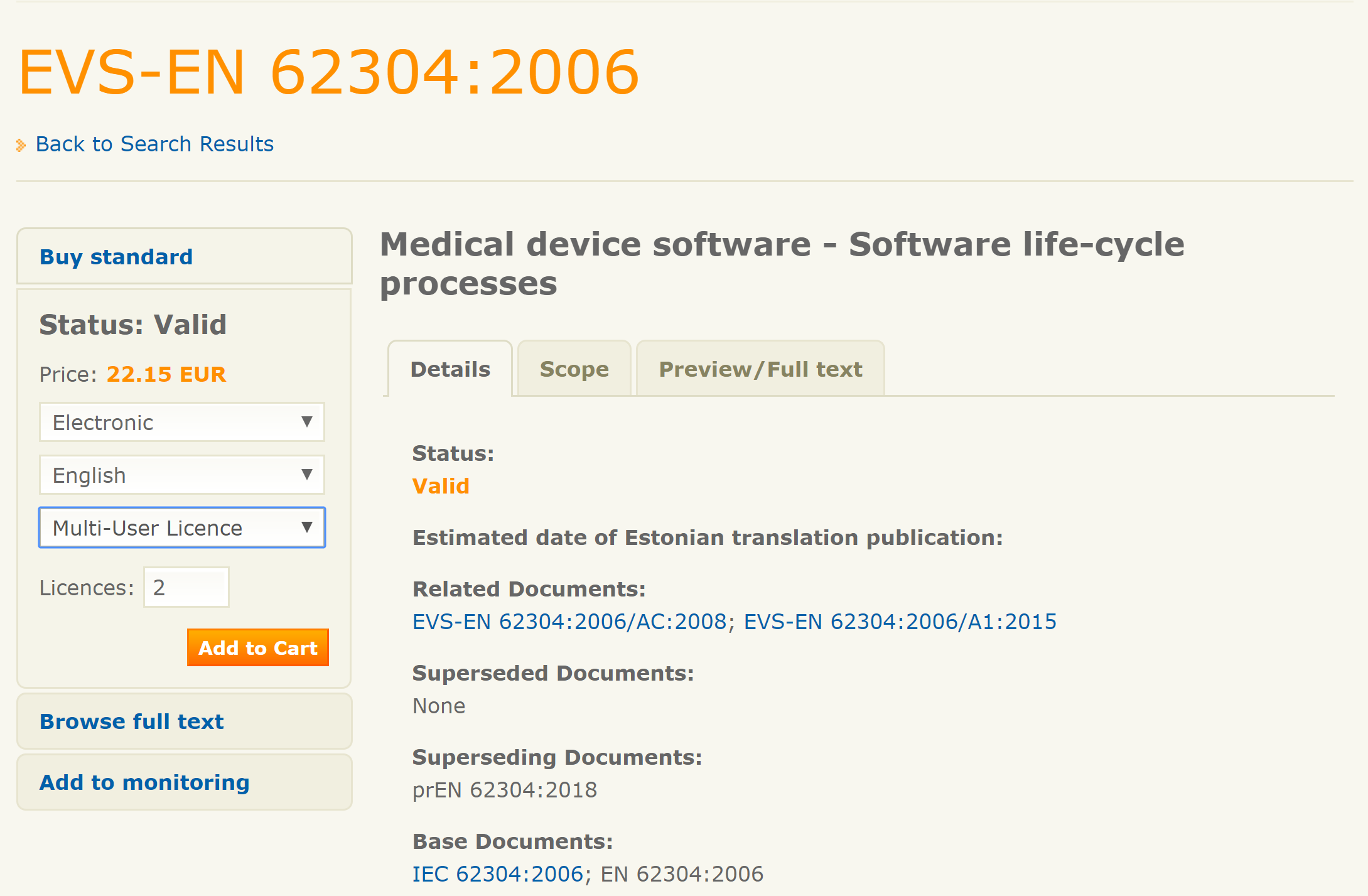
#Iso 13485 test method validation how to#
In this blog we will explain how to address this in a general matter and, as example, show you what it means when it comes to validating Matrix software. There are different standards explaining how to validate software products and some software seem impossible to validate at all. If you are not quite sure what to do for software validation, you are not alone!
